
DocetaxelCAS No.:114977-28-5
|
||||||||||
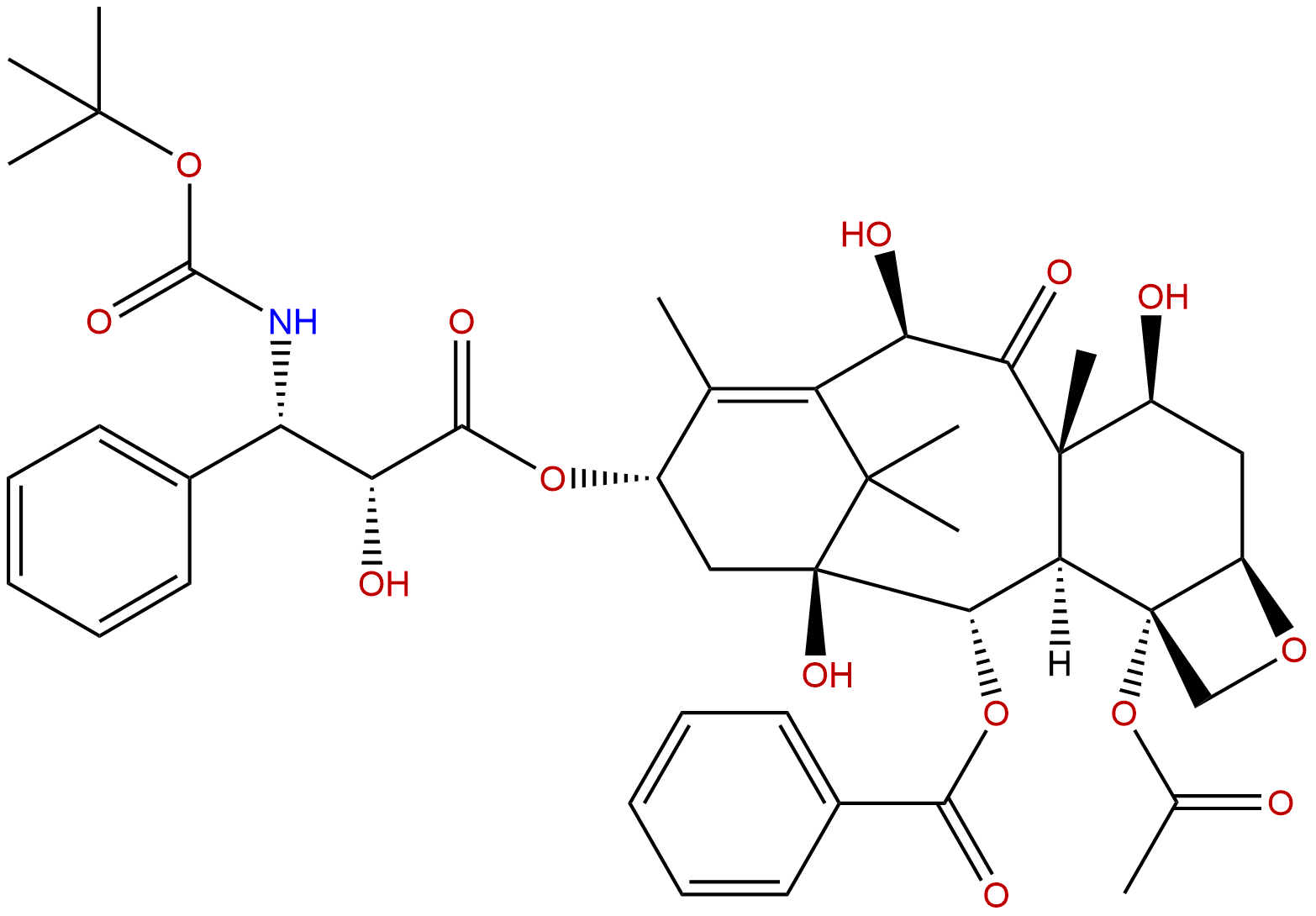 |
|
|
||||||||

| Catalogue No.: | BP0514 |
| Formula: | C43H53NO14 |
| Mol Weight: | 807.89 |
Synonym name:
Catalogue No.: BP0514
Cas No.: 114977-28-5
Formula: C43H53NO14
Mol Weight: 807.89
Botanical Source:
Purity: 95%~99%
Analysis Method: HPLC-DAD or/and HPLC-ELSD
Identification Method: Mass, NMR
Packing: Brown vial or HDPE plastic bottle
Can be supplied from milligrams to grams, up to kgs.
GMP is availale on request.
Inquire for bulk scale.
Description:
Docetaxel is an antineoplastic drug by inhibiting microtubule depolymerization, and attenuating of the effects of bcl-2 and bcl-xL gene expression.
References:
Cancer Treat Rev. 2015 Mar;41(3):262-70.
Balancing activity and tolerability of neoadjuvant paclitaxel- and docetaxel-based chemotherapy for HER2-positive early stage breast cancer: sensitivity analysis of randomized trials.
Paclitaxel and Docetaxel represent the most adopted taxanes in the neoadjuvant treatment of HER2-positive breast cancer. Questions still remain with regard to their difference in terms of activity and tolerability.
METHODS AND RESULTS:
Events for pathological complete response (pCR), severe and febrile neutropenia (FN), and severe neurotoxicity were pooled by adopting a fixed- and random-effect model. A sensitivity analysis to test for the interaction between paclitaxel and Docetaxel was accomplished. Absolute differences with 95% confidence intervals (CIs) and the number of patients needed to treat/harm (NNT/NNH) were calculated to derive the Likelihood of being Helped or Harmed (LHH). Data from 15 trials (3601 patients) were included. Paclitaxel significantly increases pCR rate by 6.8% in comparison with Docetaxel (43.4%, 95% CI 41.1-45.7% versus 36.6%, 95% CI 34.3-39.0%, p=0.0001), regardless of the chemotherapy backbone, with an absolute difference of 9% and 9.2% for anthracycline-based or free-regimens. Paclitaxel significantly improves pCR versus Docetaxel with a single HER2-inhibition by 6.7% (p=0.0012), with no difference if combined with a dual HER2-inhibition. Severe neutropenia and FN are significantly lower with paclitaxel, with an absolute difference of 32.4% (p<0.0001) and 2.5% (p=0.0059), respectively. Conversely, severe neurotoxicity is slightly higher with paclitaxel (3%, p=0.0001). The LHH ratio calculated for pCR and severe neutropenia is 2.0 and 0.7 for paclitaxel and Docetaxel.
CONCLUSIONS:
Although the activity of neoadjuvant paclitaxel and Docetaxel HER2-positive breast cancer is considered similar, the slight advantage in pCR, the significantly lower neutropenia and FN, do favor paclitaxel (in the weekly fashion) over Docetaxel, despite the slightly worst neurotoxicity.
BJU Int. 2015 May;115(5):744-52.
Docetaxel rechallenge after an initial good response in patients with metastatic castration-resistant prostate cancer.
To evaluate the benefit of Docetaxel rechallenge in patients with metastatic castration-resistant prostate cancer (mCRPC) relapsing after an initial good response to first-line Docetaxel.
METHODS AND RESULTS:
We retrospectively reviewed the records of consecutive patients with mCRPC with a good response to first-line Docetaxel [serum prostate specific antigen (PSA) decrease ≥50%; no clinical/radiological progression]. We analysed the impact of management at relapse (Docetaxel rechallenge or non-taxane-based therapy) on PSA response, symptomatic response (performance status/pain/analgesic consumption), and overall survival (OS). We used multivariate stepwise logistic regression to analyse potential predictors of a favourable outcome. We identified 270 good responders to first-line Docetaxel. The median progression-free interval (PFI) was 6 months from the last Docetaxel dose. At relapse, 223 patients were rechallenged with Docetaxel (82.5%) and 47 received non-taxane-based therapy. There was no significant difference in median OS {18.2 [95% confidence interval (CI) 16.1-22.00] and 16.8 [95%CI 13.4-21.5] months, respectively, P = 0.35}. However, good PSA response and symptom relief/stable disease were more frequent on Docetaxel rechallenge (40.4% vs 10.6%, P < 0.001 for PSA). A PFI of >6 months and added estramustine predicted a good PSA response and symptomatic response on Docetaxel rechallenge but only a PFI of >6 months predicted longer OS. Haemoglobin (<13 g/dL) and pain were associated with reduced OS. Docetaxel rechallenge increased the incidence of grade ≥3 sensory neuropathy, nail disorders and asthenia/fatigue.
CONCLUSIONS:
Docetaxel rechallenge is a management option for responders to Docetaxel with a PFI of >6 months, but did not prolong survival. Potential benefits should be weighed against the risk of cumulative toxicity.
N Engl J Med. 2004 Oct 7;351(15):1513-20.
Docetaxel and estramustine compared with mitoxantrone and prednisone for advanced refractory prostate cancer.
Mitoxantrone-based chemotherapy palliates pain without extending survival in men with progressive androgen-independent prostate cancer.
METHODS AND RESULTS:
We compared Docetaxel plus estramustine with mitoxantrone plus prednisone in men with metastatic, hormone-independent prostate cancer. Of 674 eligible patients, 338 were assigned to receive Docetaxel and estramustine and 336 to receive mitoxantrone and prednisone. In an intention-to-treat analysis, the median overall survival was longer in the group given Docetaxel and estramustine than in the group given mitoxantrone and prednisone (17.5 months vs. 15.6 months, P=0.02 by the log-rank test), and the corresponding hazard ratio for death was 0.80 (95 percent confidence interval, 0.67 to 0.97). The median time to progression was 6.3 months in the group given Docetaxel and estramustine and 3.2 months in the group given mitoxantrone and prednisone (P<0.001 by the log-rank test). PSA declines of at least 50 percent occurred in 50 percent and 27 percent of patients, respectively (P<0.001), and objective tumor responses were observed in 17 percent and 11 percent of patients with bidimensionally measurable disease, respectively (P=0.30). Grade 3 or 4 neutropenic fevers (P=0.01), nausea and vomiting (P<0.001), and cardiovascular events (P=0.001) were more common among patients receiving Docetaxel and estramustine than among those receiving mitoxantrone and prednisone. Pain relief was similar in both groups.
CONCLUSIONS:
The improvement in median survival of nearly two months with Docetaxel and estramustine, as compared with mitoxantrone and prednisone, provides support for this approach in men with metastatic, androgen-independent prostate cancer.